Navigazione principale
Navigazione principale
PhD Top Stories
Riccardo Sola
Characterization of cetacean Proline-rich antimicrobial peptides: finding novel potential antibiotics
Doctoral Programme in Molecular Biomedicine
The unwise use of antibiotics has allowed the selection and diffusion of �multi-drug resistant� microbes, which now cause thousands of deaths every year worldwide. To overcome the drug-resistances of these microbes, novel classes of antibiotics are urgently required. Novel antibiotics may come from antimicrobial peptides (AMPs), promising molecules of the immune system of most animals and plants. Most AMPs kill bacteria by binding and disrupting their envelope (�membranolytic� AMPs). Membranolytic AMPs are powerful against several bacteria but often toxic to human cells, breaking their membranes as well. However, not all AMPs act this way. Proline-rich antimicrobial peptides (PrAMPs) do not break the bacterial envelope and are generally less toxic for human cells than other AMPs. PrAMPs exploit some transporter proteins such as SbmA as �trojan horse� to enter into bacteria; once inside, PrAMPs selectively bind and block the ribosomes (the molecular machineries producing proteins), thereby killing bacteria. However, their targets are limited to the pathogens producing the �trojan horse� protein. Researchers are trying to optimize PrAMPs to broaden their microbial targets while keeping their relatively low toxicity. The study of novel PrAMPs and the relation between amino acid sequence and antibiotic properties would provide precious hints for such optimization.
The laboratory of Prof. Scocchi has been studying for years a PrAMP called Bac7(1-35), derived from a bigger peptide produced by some artiodactyls (e.g. goats, pigs), endowed with excellent antimicrobial properties. Using the amino acid sequence of Bac7(1-35) as a reference, we performed a bioinformatic research of novel �equivalent� PrAMPs in cetaceans, which are evolutionarily close to artiodactyls. This led us to discover seven cetacean PrAMPs (Fig.1), coming from different cetacean species, with different degrees of similarity with Bac7(1-35). Two peptides called Bal1 and Lip1, whose sequence most resembled Bac7(1-35), showed broader and more powerful antimicrobial activity than Bac7(1-35) itself (Fig.1).
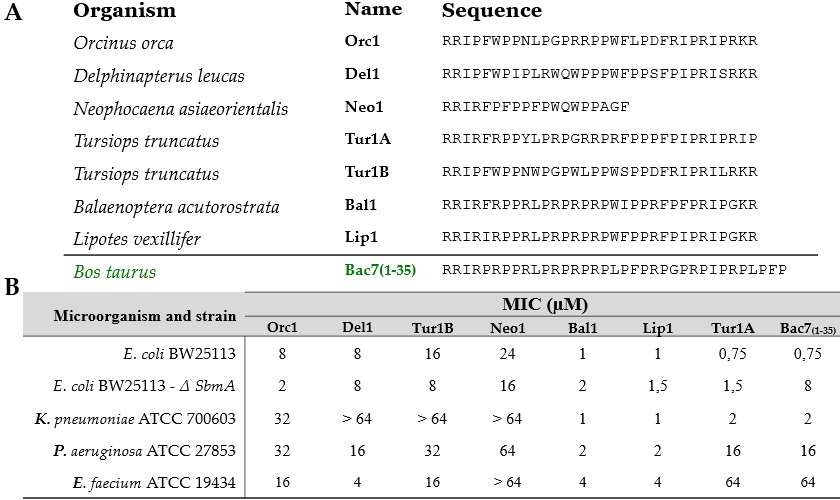
Figure 1: Sequences (A) and antimicrobial activity (B) of cetacean PrAMPs and Bac7(1-35).
Their minimum concentration inhibiting bacterial growth (MIC) was = 4 �M versus several bacteria, encompassing troublesome Gram-negative pathogens such as Pseudomonas aeruginosa and the Gram-positive Enterococcus faecium. Plus, in absence of the transporter SbmA, Bal1 and Lip1 did not lose their efficacy as much as other known PrAMPs do. Bal1 and Lip1 retained a non-membranolytic mode of action blocking the protein production (or �synthesis�) machinery (Fig.2), like Bac7(1-35). Notably, a short portion of their sequence is nearly identical in other mammalian and insect PrAMPs that block protein synthesis, suggesting a �sequence motif� related to this property. Even at doses many times their MIC, Bal1 and Lip1 were not toxic to human red blood cells and keratinocytes (not shown), showing only some cytotoxicity versus the human leukemic cells Mec-1 (Fig.2).
Figure 2: Effects of peptides on bacterial protein production(A) and toxicity (B) towards the human cell line MEC-1. A) measurement of protein synthesis by detecting the luminescence of a reference protein (luciferase). B) cell viability (MTT) assay. All results are reported as a percentage with respect to the signal of samples not treated with the peptides.
A second group of cetacean PrAMPs that we found (Orc1, Del1, Tur1B, Neo1) was quite �atypical� since they did not hamper protein synthesis (Fig.2) but rather seemed to damage bacterial membranes (not shown). Despite their mode of action, these peptides were less powerful and narrower-range than Bal1 and Lip1. Moreover, some of these peptides are non-cytotoxic also against MEC-1 cells (Fig.2). Such non-common PrAMPs surely deserve further studies. In conclusion, the study of novel cetacean PrAMPs identified two peptides with similar mode of action of Bac7(1-35) but stronger and broader action, and it also identified some unusual membranolytic peptides, less effective than Bac7(1-35) but not toxic for the tested human cells. Relating differences in the sequences of all these PrAMPs to the difference in their efficacy, toxicity and mode of action may give useful hints for the design of optimized PrAMPs, to be used as leads for developing new antibiotics.
Authors and affiliations
Riccardo Sola1, Mario Mardirossian2, Bertrand Beckert3 Laura Sanghez De Luna1 Dennis Prickett1 Alessandro Tossi1 Daniel N. Wilson3 Marco Scocchi12Dipartimento di Scienze Mediche, Universit� di Trieste, Trieste, Italy
3Istituto di Biochimica e Biologia Molecolare, Universit� di Amburgo, Amburgo, Germania
Contact
Riccardo sola, email: riccardo.sola@phd.units.itReference
R. Sola et al.Characterization of Cetacean Proline-Rich Antimicrobial Peptides Displaying Activity against ESKAPE Pathogens
Int. J. Mol. Sci. 21, 7367 (2020)
DOI: 1422-0067/21/19/7367
Informazioni aggiornate al: 15.4.2021 alle ore 09:33
Contact: Webmaster - Università di Trieste pagina curata da: Research Doctorate



